| Issue |
EPL
Volume 135, Number 3, August 2021
|
|
|---|---|---|
| Article Number | 30005 | |
| Number of page(s) | 4 | |
| Section | General | |
| DOI | https://doi.org/10.1209/0295-5075/ac1960 | |
| Published online | 18 October 2021 | |
Active adsorption performance of planetary ball milled Saudi Arabian bentonite clay for the removal of copper ions from aqueous solution
1 Department of Chemical Engineering, Faculty of Engineering, Jazan University - P.O. Box 706, Jazan 45142, Saudi Arabia
2 Department of Chemistry and Industrial Chemistry, College of Applied and Industrial Sciences, University of Bahri - Bahri, Sudan
3 Department of Chemical Engineering, Faculty of Engineering, Hadhramout University - Hadhramout, Yemen
4 Department of Chemical Engineering, Faculty of Engineering, University of Elimam Elmahadi - Kosti, Sudan
(a) imranchaudhary0@gmail.com (corresponding author)
Received: 21 March 2021
Accepted: 30 July 2021
We report the role of local bentonite clay in the removal of Cu2+ ions from aqueous solution. The fine bentonite clay powder was analysed by XRD, FTIR, SEM and DLS analysis techniques. Further, the adsorption experiments were carried out by varying many factors such as weight and size of bentonite clay, residence time, pH of the solution, stirring rate, temperature, and flow rate. The optimum conditions for effective removal of Cu2+ ions was 1 g dose of bentonite and 63 μm size of bentonite, 50 minutes of residence time and 50 °C temperature at pH 3 with a flow rate of 1 L/min. The data fitted well the Freundlich model and a maximum adsorption capacity of 61.72 mg/g has been obtained. The value of Gibbs free energy changes (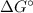 ), enthalpy changes (
), enthalpy changes (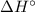 ) and entropy changes (
) and entropy changes (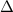 S°) were found to be
S°) were found to be 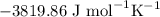 ,
, 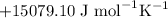 and
and 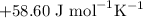 , respectively.
, respectively.
© 2021 EPLA
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


