| Issue |
Europhys. Lett.
Volume 46, Number 3, May I 1999
|
|
|---|---|---|
| Page(s) | 382 - 388 | |
| Section | Condensed matter: electronic structure, electrical, magnetic and optical properties | |
| DOI | https://doi.org/10.1209/epl/i1999-00100-3 | |
| Published online | 01 September 2002 | |
Effect of charge state on bonding in fulleride polymers
Department of Physics and Measurement Technology-IFM, Linköping University
S-581 83, Linköping, Sweden
Received:
4
November
1998
Accepted:
17
February
1999
Single-bonded fulleride polymers of one-, two- and three-dimensional phases
have been investigated theoretically using the Hückel model.
Parameters for the sp3 hybridized carbon atoms forming the bonds have been
found by fitting the energy spectrum and bond orders of the bonding atoms
of a single-bonded 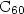 dimer to the results of Valence Effective Hamiltonian calculations.
Our calculations strongly support previous results and lead to the general
conclusion that each electron added to
dimer to the results of Valence Effective Hamiltonian calculations.
Our calculations strongly support previous results and lead to the general
conclusion that each electron added to 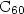 stimulates
the creation of one single bond to a neighbouring
stimulates
the creation of one single bond to a neighbouring 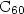 anion. Furthermore, despite the fact
that the intermolecular bonds are formed between two sp3 hybridized carbon atoms,
all the polymer phases exhibit extended states.
anion. Furthermore, despite the fact
that the intermolecular bonds are formed between two sp3 hybridized carbon atoms,
all the polymer phases exhibit extended states.
PACS: 71.20.Tx – Fullerenes and related materials; intercalation compounds / 31.15.Ct – Semi-empirical and empirical calculations (differential overlap, Hückel, PPP methods, etc.)
© EDP Sciences, 1999
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


